
Retail

Although FDA concluded OTC oral phenylephrine is ineffective as a nasal decongestant in doses up to highest studied level, federal preemption precludes challenges to agency’s regulation of labeling for phenylephrine and other Rx and OTC drugs, according to recent decision in New York federal court.

Firm’s future, as a separate business nearly 90% owned by Bausch Health, or as a standalone with different ownership or one not controlled by a majority owner, claims high profile in analysts’ comments and predictions. B+L is in the driver’s seat, they say.

Reckitt backs up prediction for strong sales during current quarter with advertising featuring a beauty queen and a line extension for Mucinex, one its strongest US consumer health product lines.

Requests for “enforcement actions are not within the scope of FDA’s citizen petition procedures,” CDER says, rejecting petition dosing device firm Parenteral Technologies submitted as it prepares for workshop on Pediatric Research Equity Act requirements for OTC NDA sponsors.

UK pharma also reaches agreement in principle, subject to DoJ approval, to pay $70m to resolve a whistleblower complaint filed by Valisure, the testing lab which in 2019 raised concerns about a potential link between the use of drugs containing ranitidine, a histamine-2 blocker, and cancer.

Regenerative Processing replaces nozzle to prevent backflow for its Regener-Eyes drops but FDA warning states numerous questions about sterility at the firm’s plant and about its procedures and systems for preventing microbial contamination.

After launching Retaine MGD Advanced, OcuSoft says a release by Bruder Healthcare referenced Retaine MGD trademark and statements from a previous OcuSoft announcement about the original product attributed to an optometrist. B+L, Rohto brand and homeopathic firm Relief Products also make US OTC eye care space moves.

Jessica Spence joins as North American president while CFO Dan Sullivan becomes COO and Francesca Weissman, finance and business strategy SVP, adds CFO to her title in latest executive office changes by the marketer of brands including Schick men's and women’s shaving, Playtex, Stayfree, Carefree and o.b. feminine care and Banana Boat and Hawaiian Tropic sun care.

Nonbinding forecast, fourth for the program, includes risks associated with codeine-containing cough medicine as a topic FDA will include in ongoing evaluation of GRASE for pediatric cough cold drug products marketed under monograph for cold, cough, allergy, bronchodilator and anti-asthmatic OTC drugs.

Mucinex Sinus saline nasal spray is designed to treat sinus-related symptoms as “first-ever drug-free saline product” with a dual nozzle offering two spray settings, marketer Reckitt-Benckiser says.

Council for Responsible Nutrition contends New Jersey bill, which Assembly Health Committee amended with substantial language to make restrictions more stringent on 23 September, is targeted as inaccurately as restrictions in New York effective in April.
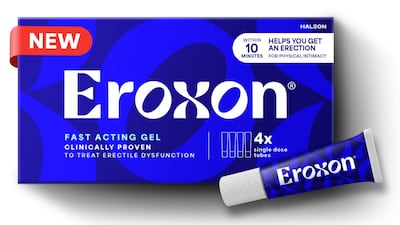
October will see the bricks-and-mortar launch of the first FDA-approved OTC treatment for erectile dysfunction in the US by Haleon. HBW Insight catches up with the company's North America president Lisa Paley to find out what Haleon has planned for Eroxon's arrival in stores.

Following Energy and Commerce Health Subcommittee hearing about FDA’s human food and tobacco programs on 10 September, gap between what the trade groups, committee leadership and the FDA each want more of doesn’t appear to be shrinking.

FDA approves Flumist consumers can use at home for prevention of flu caused by virus subtypes A and B in individuals 2 through 49 years old in a proposal submitted by AstraZeneca’s MedImmune.

In addition to commentary and information about the OTC drug, dietary supplement and cosmetic industries, speakers offered candid remarks on subjects well known across the consumer health industry or well recognized by consumers.

Green Pharmaceuticals’ SnoreStop Nasal Spray, previously marketed as “NasoSpray,” still is available even though agency officials on multiple occasions for a month recommended a recall after an April inspection found “gross microbial contamination” in one lot.

Bayer’s Midol and nonprofit Period. provide PeriodTalk campaign for awareness of the gaps in period education; “The M Factor” film coming day before World Menopause Day in October; Exeltis USA’s Blues Away provides postpartum mood support; and Doctor's Best Women's Collection includes menopause, sleep, digestive, heart and hair/skin/nails support products.

Joint letter on scheduling for proceedings before a trial CRN’s attorneys and state AG’s office recently submitted in New York federal court includes, among other information, a statement that both the trade group and the state anticipate a settlement could be reached.

Court order dismisses claims against Perrigo and Joseph Papa, its former CEO, with prejudice. Settlement also bars any claim against plaintiffs and class members, “whether arising under state, federal, common or foreign law,” alleging liability for losses.

“Control of Nitrosamine Impurities in Human Drugs” guidance, FDA’s third on the subject, adds nearly 20 pages to 2021 guidance document it revises with more extensive discussions of each topic included in the earlier guidance.